Who We Are
Process Design Solutions (PDS) provides client services and engineering solutions to the life science industry.
Our Story
“Unlike many other engineering firms, Process Design Solutions (PDS) focuses our process engineering efforts on product quality and patient safety. Since 2009, Quality by Design (QbD) and its principles and concepts have driven our mission, vision, and values. PDS has invested in technology and innovation to drive QbD as a focal point of our core business. We have integrated this approach into our service model to deliver projects ‘Right First Time.’”
—Paul Stanovich, Managing Partner
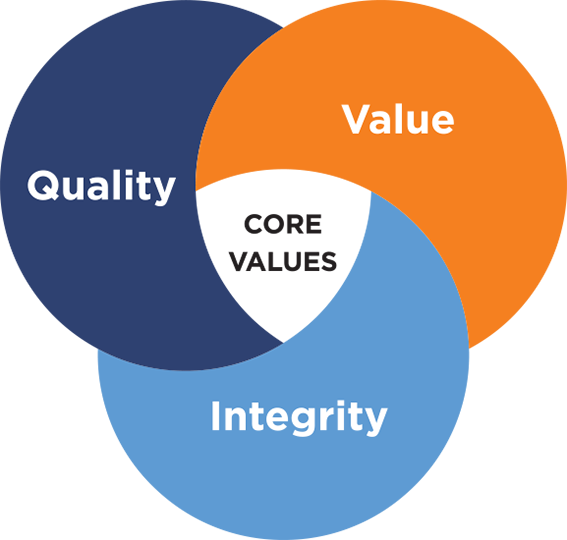
Our Values
As trusted advisers, we strive to provide the highest possible value in everything we do.
This means we ensure…
Quality in what we deliver, first and foremost
Value that exceeds our customers expectations
Integrity which is demonstrated every day
Our Approach
Our unique approach applies the concepts of QbD to improve all aspects of the process engineering function.
Our innovative solutions combined with deep subject matter expertise help companies deliver projects right the first time.
How We Do This
Listen. Plan. Deliver. By strategically focusing design efforts on product quality and patient safety, we extend the principles of QbD into the process engineering space for manufacturing systems, helping clients appropriately apply the principles of...
Quality Risk Management (QRM) via ICH Q9
Engineering Change Management (ECM) as a supporting process
Quality Systems (QS) to manage impact to critical aspects
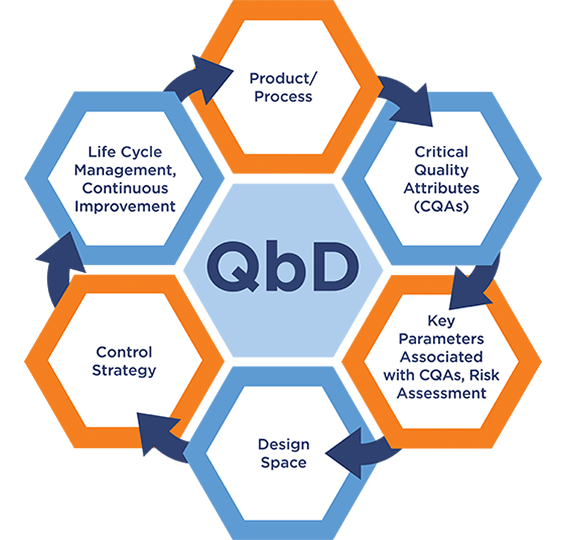
Quality by Design (QbD, ICH Q8) is the systematic approach to development that begins with predefined objectives and emphasizes product and process understanding and process control, based on sound science and quality risk management.
Listen. Plan. Deliver. Success By Design.
Services
PDS is a trusted partner providing clients expertise in:
- Project & Process Engineering
- Automation Engineering
- Commissioning & Qualification
- Engineering Deliverables
- Project Management
- Program Management
Supporting all project phases for new and existing facilities.
- Conceptual & Basic Design
- Detailed Design & Project Execution
- Fixed Equipment & Single-Use Systems
- Tech Transfer & New Product Introduction
- Engineering Operational Support

Automation engineering in all phases for new, existing and multi-product facilities.
- Conceptual Design for Control Systems
- Specification, Design & Implementation
- Alarm Design & Rationalization

CQV support for all phases for new, existing and multi-product facilities.
- Verification Strategy
- Start-up & Engineering Testing
- Commissioning
- Qualification
- Computerized Systems Validation

Generate Engineering Deliverables Accurately & Efficiently
With proEngQbD, engineering deliverables can be generated quickly and updated automatically.
- Process Flow Diagrams (PFDs)
- Piping & Instrumentation Diagrams (P&IDs)
- Fixed & Single-Use Equipment Capabilities
- System & User Requirement Specifications
- Design Review & Facility Fit Assessments
- Risk Assessments


- Providing effective project management representing our clients as owner reps from conception through closure for CAPEX and OPEX projects

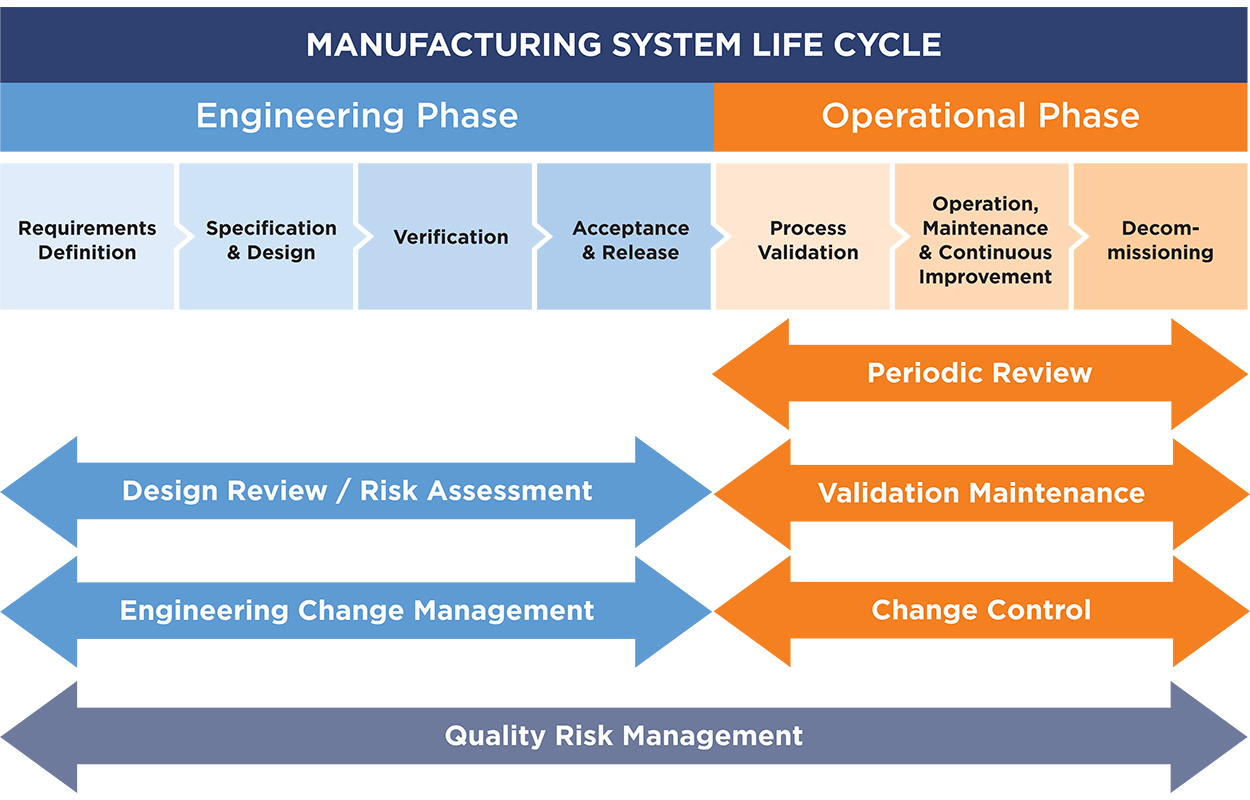
All phases of planning and implementing ASTM E2500 to manage all elements of the engineering and operational phases of the manufacturing system lifecycle and QbD.
- ASTM E2500 Planning & Implementation
- Change Management
- Design Review & Risk Assessment
- QRM Consulting and Training

Accelerate Your Projects with a Digital Solution for Engineering Deliverables
proEngQbD offers a systematic approach that enhances the efficiency and accuracy of engineering work.
View SolutionPlay VideoproEngQbD is powered by Siemens COMOS Industry Software.


A Single Point of Truth for Engineering Data
An innovative solution for engineering Information Management (EIM) for FDA and other regulated industries. Providing a more flexible and agile approach for developing and managing engineering data.
View SolutionPlay VideoproEngQbD is powered by Siemens COMOS Industry Software.

COMOS -
Making Data Work
Better quality decision-making throughout the plant’s entire lifecycle

COMOS provides the process industry with a seamless flow of information based on a globally consistent database – integrated across all disciplines and all phases of a plant's lifecycle. This is possible thanks to the systematic application of object orientation. This means: In COMOS, all data relating to the same component form a unit – an object. Changes to object specifications are stored in the central COMOS database so that the updated data is available everywhere and at all times. Plant engineers and operators can access data that is always up-to-date – in real time and independent of time zones. That creates the basis for maximum decision-making reliability – and, thereby, for considerably greater productivity.
COMOS benefits at a glance:
- Integrated and consistent plant management
- Flexible application options in all disciplines and all lifecycle phases of a plant project
- Optimal integration and coordination of all plant subsections involved in planning and operation
- Reduction of process cycle times through close interaction between the engineering and operating phases
- Reduced data complexity through central storage of all plant information enables consistent access to all object-related data for any user
- Open system architecture for perfect adaptation to operation-specific requirements and integration of third-party systems into existing IT landscapes
The high flexibility of COMOS solutions can be individually tailored to your specific needs – and that's where we come in. As a company with extensive industry-specific know-how, we at apply all of our expertise to benefit your plant. We offer comprehensive consulting and technical support for customizations as well as training courses for COMOS users – for even more performance, quality and local presence.
More InformationSoftware Solutions for Optimized Plant EngineeringCOMOS - Making Data Work
Better quality decision-making throughout the plant’s entire lifecycle
COMOS provides the process industry with a seamless flow of information based on a globally consistent database – integrated across all disciplines and all phases of a plant's lifecycle. This is possible thanks to the systematic application of object orientation. This means: In COMOS, all data relating to the same component form a unit – an object. Changes to object specifications are stored in the central COMOS database so that the updated data is available everywhere and at all times. Plant engineers and operators can access data that is always up-to-date – in real time and independent of time zones. That creates the basis for maximum decision-making reliability – and, thereby, for considerably greater productivity.
COMOS benefits at a glance:
- Integrated and consistent plant management
- Flexible application options in all disciplines and all lifecycle phases of a plant project
- Optimal integration and coordination of all plant subsections involved in planning and operation
- Reduction of process cycle times through close interaction between the engineering and operating phases
- Reduced data complexity through central storage of all plant information enables consistent access to all object-related data for any user
- Open system architecture for perfect adaptation to operation-specific requirements and integration of third-party systems into existing IT landscapes
The high flexibility of COMOS solutions can be individually tailored to your specific needs – and that's where we come in. As a company with extensive industry-specific know-how, we apply all of our expertise to benefit your plant. We offer comprehensive consulting and technical support for customizations as well as training courses for COMOS users – for even more performance, quality and local presence.
More InformationSoftware SolutionsClients

















